A little story about drug development

A little story about drug development
Drugs have existed long before technologies were available to characterize their chemical structures let alone their biological targets. From early discoveries that relied on nature’s resources to modern large-scale screening approaches, this blog post provides a short overview of the history of drug development.
November 22, 2025
Over the last centuries drug development has taken a different shape. Initially, compounds with therapeutic effects were identified in nature by chance. While nature continues to be a rich resource to inform drug development today, new systematic approaches have proved to be more successful than relying merely on accidental or serendipitous discoveries. This blog post briefly lists famous serendipitous discoveries resulting in therapeutics still in use today, introduces the drug receptor theory, and describes the concepts of forward and reverse pharmacology, the latter being applied more recently and nowadays strongly capitalizes on genomic data stored in large-scale biobanks.
Click to view the chemistry story of aspirin
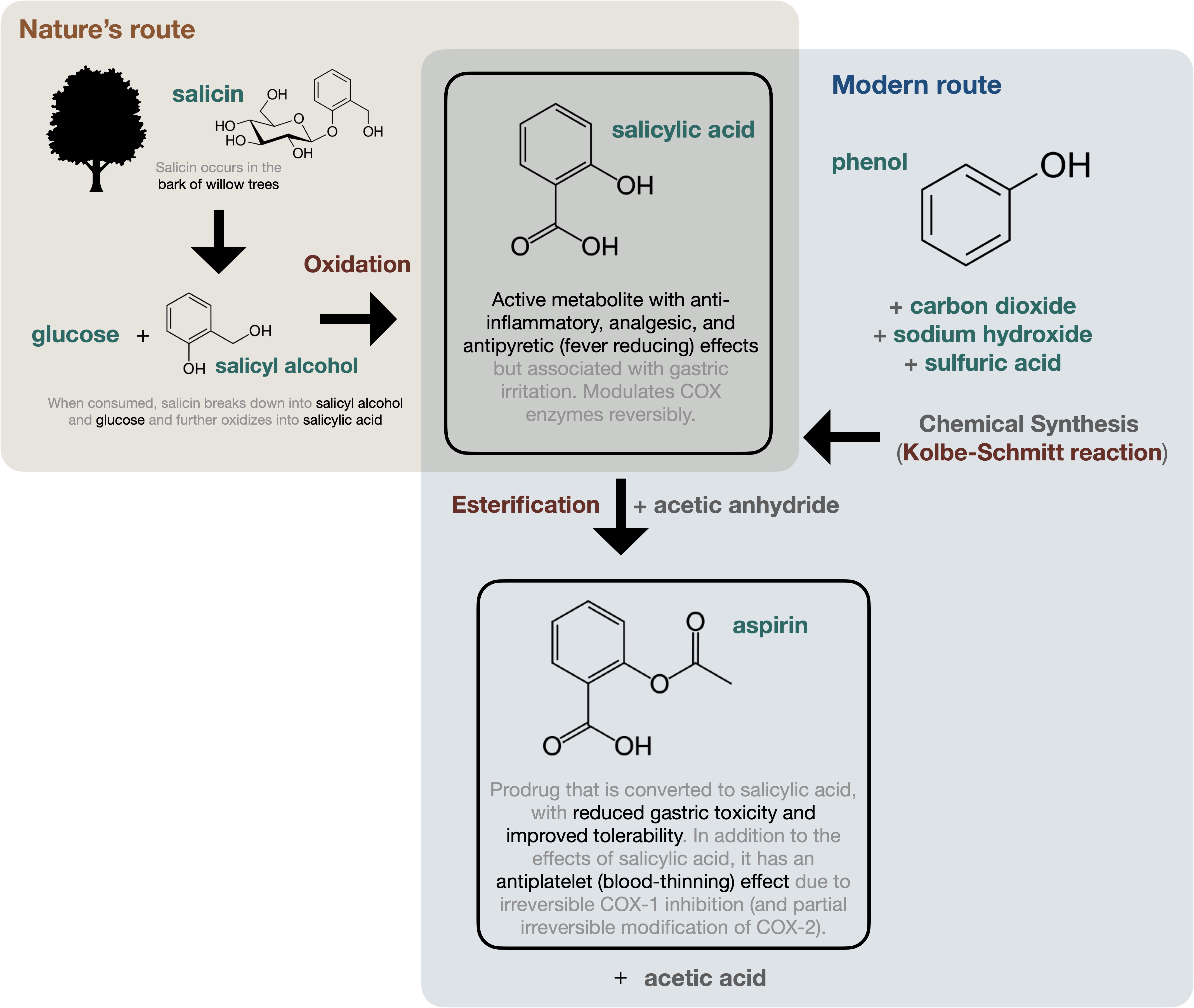
Table of Contents
- Early stages - serendipitous discoveries
- Drug receptor theory
- Systematic drug screening
- Forward and reverse pharmacology
- Genomics for target discovery
- Concluding remarks
- References
Early stages - serendipitous discoveries
Drug discovery has been around for a long time thanks to Nature’s rich resources, and some successful drugs still used in the clinics today have emerged over 200 years ago. The following drugs are examples of serendipitous discoveries, or accidental discoveries, that were made in the 19th and 20th centuries.
-
Morphine.
Morphine isolated from opium in the early 1800s can be considered as the first modern, pharmaceutical medicine as it was the first time an active pharmacological compound has been isolated from natural sources in a pure state. -
Chloral hydrate.
Chloral hydrate is one of the first synthetic drugs. Justus von Liebig prepared this drug in 1832 and showed that it converted into chloroform and formic acid when put into an alkaline solution. Based on this observation, Oscar Liebreich a German physician thought that this effect could occur directly in the blood and serve as an anesthetic such as chloroform. While no chloroform results from the degradation of chloral hydrate in blood, chloral hydrate was found to be a sleeping aid (hypnotic) and was used as therapeutics from 1869 onwards and is still in use today. -
Lithium.
The use of lithium as mood disorder treatment was the result of false theories about the underlying causes of mood disorders. Originally, it was tested for the treatment of gout since it was found that lithium carbonate could dissolve urate stones and gout, a disease characterized by urate crystal deposits in joints and elevated uric acid levels. Mood disorders were thought to be a form of cerebral gout which prompted William Hammod to use lithium as a treatment. Based on the same logic, Carl Lange, a Danish neurologist, used lithium to treat mood disorders in 1896. Since lithium can be toxic, it took a few decades for lithium to be rediscovered and employed safely in the 1960s with the employment of the flame spectrophotometer, an analytical instrument that can measure the concentration of certain metal ions, to monitor blood levels. -
Penicillin.
The famous discovery of penicillin by Alexander Fleming in 1928 resulted in the first antibiotic, providing the first effective weapon against bacterial infections. This life-changing discovery earned him the Nobel Prize in Physiology or Medicine in 1945. While conducting research on staphylococci, he noticed that one of his culture plates had become contaminated by a fungus, around which there was a bacteria-free zone.
Drug receptor theory
In the early 20th century, Paul Ehrlich, who is considered the founder of immunology, introduced the receptor theory according to which receptors, initially called side chains, associated with either cells or more generally located in the bloodstream are able to bind to distinct toxins. His ``magic bullet” concept states that drugs selectively target disease-causing agents while sparing healthy tissues.
In the mid-20th century, the drug receptor theory evolved into the receptor-occupancy theory which was further refined to account for the intrinsic activity of drugs (i.e., their ability to induce an effect after binding), spare receptors (i.e., maximal drug response can be obtained with less than all receptors occupied) and binding affinities. The distinction of the α and β receptors eventually led to the introduction of propranolol, the first clinically useful β-receptor blocker.
Systematic drug screening
In the late 20th century, advances in biochemistry and molecular biology such as X-ray crystallography, NMR greatly and protein–ligand co-crystallography improved the understanding of the structure and function of proteins and molecules. Recombinant DNA technology played a major role in studying the pharmacology and function of GPCRs, G protein-coupled receptors the largest family of membrane-bound receptors that represents targets of a third of FDA-approved drugs. These cell surface receptors can detect chemical signals in a highly selective way and transmit the signal to generate intracellular responses. Subsequently, GPCR and other high-throughput assays were screened for drug interactions where the development of combinatorial libraries containing millions of potential drugs marked another major milestone in the early 1990s. Screening libraries include not only synthetic molecules developed through such combinatorial synthesis approaches where drug-like compounds with various rings and functional groups can be assembled rapidly, but also natural compounds.
Forward and reverse pharmacology
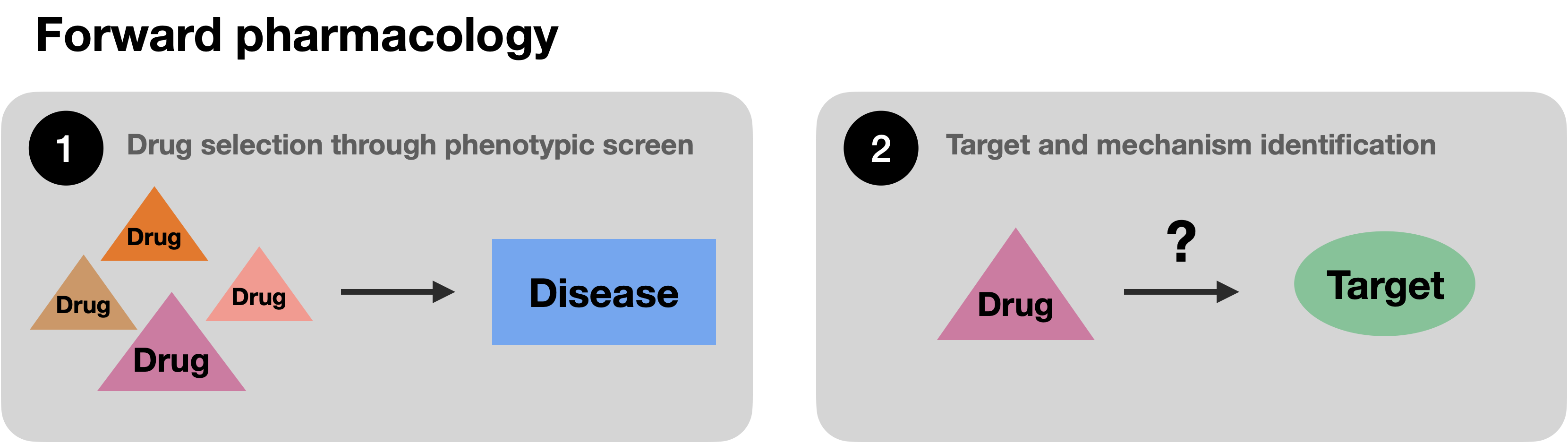
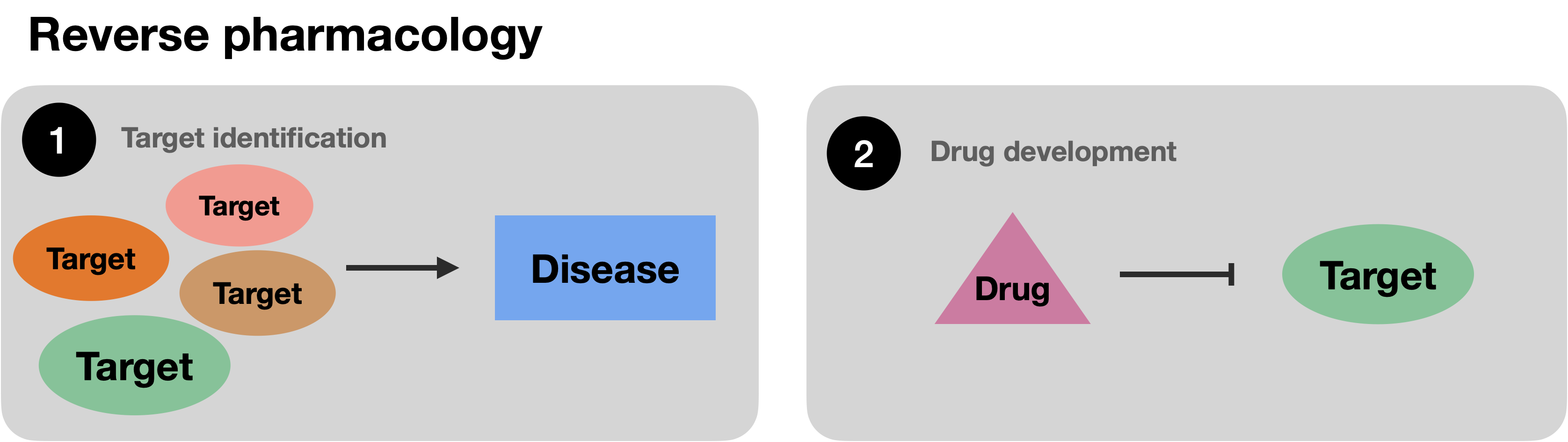
Over time, drug discovery has transitioned from forward pharmacology, or phenotypic screening, where drugs with often unknown molecular mechanisms of action have been selected based on their therapeutic impact, to reverse pharmacology, or target-based screening where proteins identified to play a role in disease are tested for interaction with small molecules or biologicals such as monoclonal antibodies.\
Genomics for target discovery
A typical example of modern reverse pharmacology is the use of human genetics for target discovery. Everyone carries different genetic variants, some of which can be protective against a disease or, conversely, put you at higher risk. The genomic revolution was accelerated by the Human Genome Project (1990–2003), which brought the promise of transforming the field and helping to identify disease-associated genes. Now, contrary to early expectations, genome-wide analyses have shown that for many diseases there are not just a few genes associated with disease risk, but up to hundreds, typically each with very small effects, meaning large sample sizes are required to detect them. Not only the polygenicity of diseases, but also the fact that many risk signals lie in the non-coding (rather than coding) part of the genome, makes genetic target identification challenging. Nonetheless, drug targets with human genetic support are enriched for successful outcomes often cited as roughly two-fold—compared with targets without such support. Said more simply, when considering two potential target genes for an indication, the one supported by human genetics (for example through a Mendelian form of disease or large-scale population studies) is about twice as likely to reach approval as one without such support.
Concluding remarks
Drug discovery has come a long way from nature’s rich resources, which continue to be investigated today. The field is highly interdisciplinary, combining chemistry, biology, biochemistry, and more recently genomics, among other disciplines, all of which continue to play a part in drug development. The complexity of drug development is reflected in the high failure rates of clinical trials as well as the many diseases for which there are no effective treatments. Over time, drug development has become more systematic, both in terms of the variety of chemical and biological compounds tested through the generation of large libraries, and in terms of understanding disease mechanisms and thereby selecting targets in a more informed manner. There has been tremendous technological progress in recent years, which will hopefully translate into higher success rates and shorter development times.
Want to read more?
For further reading on the use of human genetics for drug target identification, have a look at the pioneering publication by Nelson et al. 2015; as well as a follow-up paper led by the same author Minikel et al. 2024. For a more general review on genetics for drug development have a look at the review by Chen et al. 2025. If you are interested in the technicalities of methods that use human genetics and molecular networks for target discoveries, you may want to check out my publication on that topic.
References
References (click to expand)
-
Montinari MR, Minelli S, De Caterina R. The first 3500 years of aspirin history from its roots–A concise summary. Vascular pharmacology. 2019 Feb 1;113:1-8.
-
Jones AW. Early drug discovery and the rise of pharmaceutical chemistry. Drug testing and analysis. 2011 Jun;3(6):337-44.
-
Ban TA. The role of serendipity in drug discovery. Dialogues in Clinical Neuroscience. 2006 Sep 30;8(3):335-44.
-
Strebhardt K, Ullrich A. Paul Ehrlich’s magic bullet concept: 100 years of progress. Nature Reviews Cancer. 2008 Jun;8(6):473-80.
-
Maehle AH, Prüll CR, Halliwell RF. The emergence of the drug receptor theory. Nature Reviews Drug Discovery. 2002 Aug 1;1(8):637-41.
-
Hill SJ. G‐protein‐coupled receptors: past, present and future. British Journal of Pharmacology. 2006 Jan;147(S1):S27-37.
-
Gershell LJ, Atkins JH. A brief history of novel drug discovery technologies. Nature Reviews Drug Discovery. 2003 Apr 1;2(4):321-7.
-
Swinney DC, Anthony J. How were new medicines discovered?. Nature Reviews Drug discovery. 2011 Jul;10(7):507-19.
-
Nelson MR, Tipney H, Painter JL, Shen J, Nicoletti P, Shen Y, Floratos A, Sham PC, Li MJ, Wang J, Cardon LR. The support of human genetic evidence for approved drug indications. Nature genetics. 2015 Aug;47(8):856-60.